Easy storage1


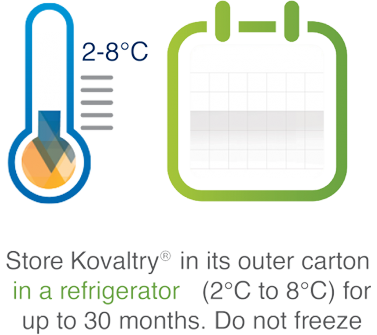
For important safety and use information, please see summary of product characteristics or country-specific prescribing information.
Recommendations
References
- Kovaltry Summary of Product Characteristics. Leverkusen, Germany: Bayer; 2017. Return to content





